Welcome to our exploration into the exciting world of paper batteries – a groundbreaking innovation that’s set to revolutionize the field of energy storage. While seemingly simple in concept, this technology is nothing short of extraordinary in its potential implications. In this blog post, we’ll dive deep into understanding the working mechanism, construction, advantages, and widespread applications of paper batteries. So buckle up, and let’s embark on this enthralling journey together!
Paper batteries, as the name implies, are batteries that leverage the thin, flexible properties of paper. They are not just a concept or a prototype but a real-world innovation that’s currently under advanced stages of development. These batteries are incredibly lightweight and flexible and can be cut or folded without losing their functionality. The “paper” in the paper battery is infused with an electrolyte and carbon nanotubes that act as the battery’s electrodes. This unique combination allows the paper battery to conduct electricity, store energy, and even act as a supercapacitor in some cases. Because they’re thin and flexible, paper batteries hold enormous potential for use in a vast range of applications, from wearable electronics to large-scale power grids.
In an era of growing energy needs, the importance of innovative energy storage solutions cannot be overstated. Breakthroughs like paper batteries come at a critical juncture, promising not only increased energy efficiency but also a greener and more sustainable way of storing power. As the push for renewable energy sources continues, the demand for efficient, compact, and flexible energy storage solutions is on the rise. Traditional batteries, with their bulky size and limited lifespan, often fall short of these requirements. Paper batteries, on the other hand, with their remarkable flexibility, lightweight, and potential for high energy storage, present a promising alternative. This could lead to advances in a range of sectors – from consumer electronics to renewable energy storage, opening up a world of possibilities.
Paper batteries = Paper + Carbon nanotubes(CNT)
1. What are carbon nanotubes (CNT)?
Carbon nanotube is an allotrope of carbon. Allotropy is nothing but different structural modifications of an element. For example, the following image shows the different allotropes of carbon. At the heart of a paper battery lies its core component – carbon nanotubes or CNTs. These are essentially sheets of graphite, an allotrope of carbon, rolled into tubes with diameters measuring mere nanometers (billionths of a meter). Despite their minuscule size, CNTs exhibit remarkable properties. They are incredibly strong – about 100 times stronger than steel – yet astoundingly lightweight. They also boast excellent conductivity of both electricity and heat, outperforming copper on both fronts. This combination of strength, lightness, and conductivity allows CNTs to store and conduct electrical energy efficiently, making them integral to the functioning of paper batteries.
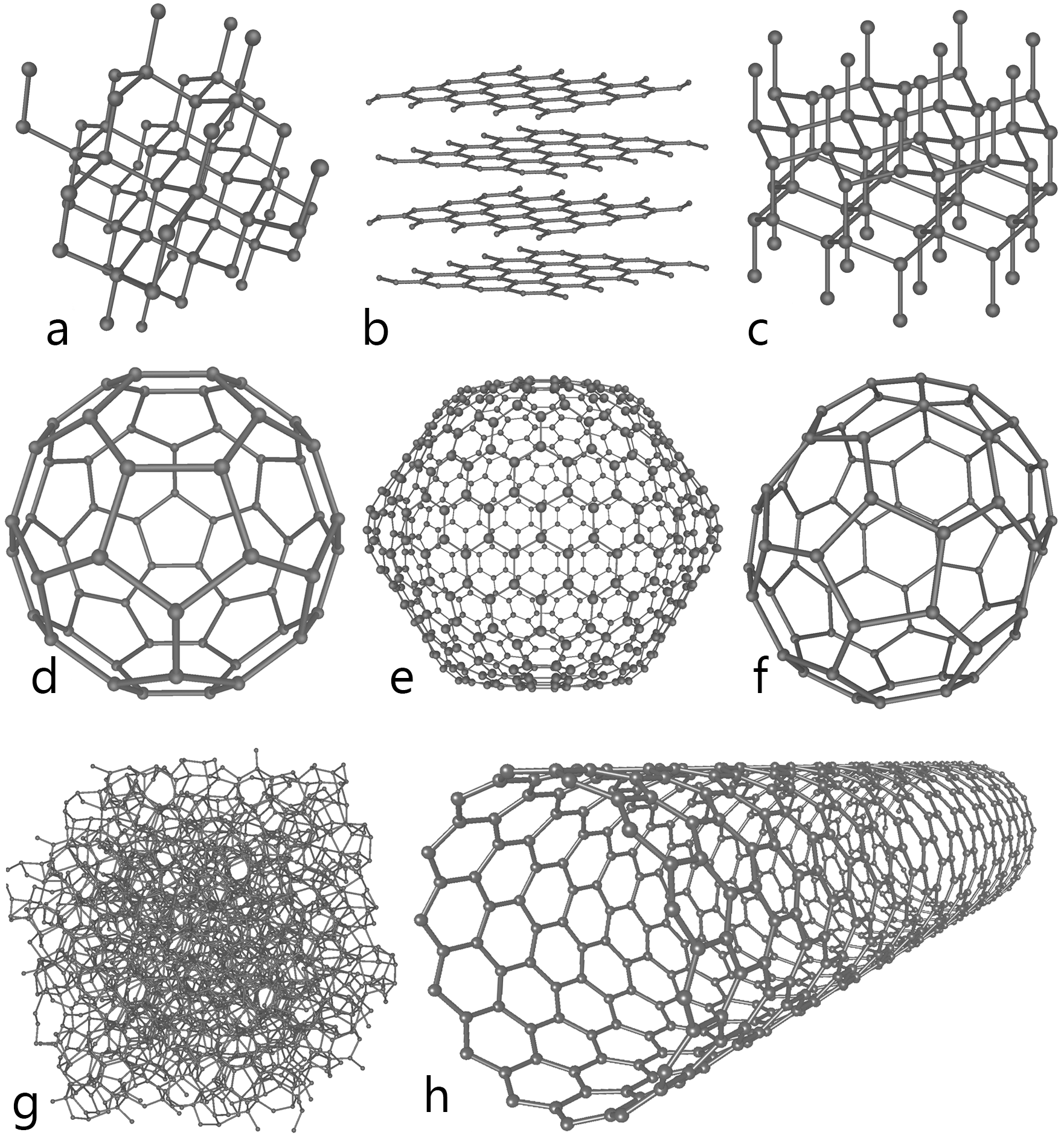
- A-Diamond
- B-Graphite
- C-Lonsdaleite
- D-Buckminsterfullerene
- E-C540
- F-C70
- G-Amorphous
- H-carbon nanotube
As you can see from the figure all the allotropes have different arrangement of atoms. For example, in case of diamond, the hardest element found on the earth, every single carbon atom is linked or bonded (covalent bond) with every other 4 atoms of carbon. Likewise, carbon nanotubes are a one of the allotropes of carbon where every carbon atom is linked with every other three carbon atoms and also form a cylindrical structure. Carbon nanotubes are man-made. The graphite and diamond are the naturally occurring allotropes of carbon.
2. What are Paper Batteries?
Paper batteries are a type of flexible, ultra-thin energy storage device composed of cellulose (paper) and infused with aligned carbon nanotubes. The term ‘paper’ in ‘paper batteries’ is used to denote the thin, flexible nature of the device, similar to a sheet of paper. What sets these batteries apart is their ability to utilize the combination of carbon nanotubes and cellulose to conduct electricity. This provides a dual benefit: the mechanical strength of carbon nanotubes and the advantage of being environmentally friendly due to the use of cellulose. They can be bent, folded, or cut into any shape without loss of integrity or efficiency, offering tremendous scope for miniaturization and adaptability into a wide range of applications.
3. Working of Paper Batteries
Cathode : Carbon nano tube (CNT)
Anode : Lithium
Electrolyte : Bio electrolyte – blood, sweat, urine, etc
Separator : Paper
Anode and cathode are the metal electrodes in the battery. The electrodes are placed in the battery in such a way that, they are in contact with the electrolyte. When an external load connected to the battery circuit is closed, current begins to flow because of the electro chemical reactions occurring inside the battery. Oxidation and reduction are the two main electro chemical reactions happening in any batteries. Oxidation takes place at anode and reduction takes place at cathode. Anode loses electrons to the ions from the electrolyte to form a compound and hence is said to be positively charged. Cathode gains electron from the electrolyte to form a compound and hence is negatively charged.
ALSO CHECK OUT OUR OTHER ARTICLES
- The Difference Between Science, Engineering, and Technology
- How To Convert Load To Gcash (Step-by-step Guide in 2023)
- How to Increase tweets impression using Useviral
- Step-by-step Guide on How to Transfer Google Drive to Another Account
- Complete Guide on How to Cash-in in GCash – 2023
4. What is the Role of the Separator?

The separator serves a critical function in paper batteries. This component is generally a thin film placed between the battery’s two electrodes, anode and cathode. Its primary role is to prevent direct contact between these electrodes, thus averting a short circuit. However, the separator is permeable to ions, allowing them to pass through it. This means while it physically isolates the electrodes, it doesn’t obstruct the ion flow, essential for the electrochemical reaction that generates electricity. The separator’s design and material composition are pivotal in ensuring efficient ion transmission and overall battery performance. The separator separates the anode and cathode. The batteries have to be very compact in size for the ease of use and hence the cathode and anode are closely packed. The separator acts as a barrier preventing the anode and cathode from touching each other. When they come in contact there will be no flow of current in external circuit.
5. Construction of Paper Batteries

The construction of paper batteries involves layering sheets of cellulose, embedded with carbon nanotubes, to form the anode, cathode, and separator. The carbon nanotubes infused within the cellulose sheets provide high electrical conductivity and mechanical strength. The sheets are soaked in an electrolyte solution, allowing the cellulose to absorb it and become ionic conductive. This absorption by cellulose transforms the paper into a separator, allowing ions to move freely while preventing a short circuit. The anode and cathode are made from a metal, usually lithium, and a lithium compound, typically lithium cobalt oxide. These layers are assembled in a sandwich-like structure, generating an ultra-thin, flexible, and lightweight paper battery. The flexibility of this device is one of its unique selling points, enabling it to be adjusted and tailored according to the needs of various applications.
- A common xerox paper of desired length is taken
- A specially formulated ink (with CNT) is spread over the paper
- On the other side a thin film of lithium is laminated.
- Aluminium rods are connected for carrying current.
6. Advantages of Paper Batteries

Paper batteries offer numerous advantages that make them a promising innovation in the field of energy storage. Firstly, they are extremely thin, lightweight, and flexible, which makes them highly adaptable to a variety of applications, especially for devices that demand portable and compact power sources. They can be cut, folded, or rolled without losing their functionality, providing flexibility in design and usage.
Secondly, paper batteries are environmentally friendly. They are composed of cellulose, a common and easily renewable material, and carbon nanotubes, reducing the reliance on toxic heavy metals typically used in traditional batteries. This composition allows them to be biodegradable, posing minimal environmental impact when they reach the end of their lifecycle.
Thirdly, they boast a high energy-to-weight ratio. The presence of carbon nanotubes in the cellulose matrix provides a large surface area for energy storage, leading to higher power density. This means paper batteries can potentially store the same amount of energy as a conventional battery, but at a fraction of the weight.
Lastly, the ability of the cellulose to absorb various types of electrolytes means paper batteries can be powered by different liquids, including biological fluids, expanding their possible applications in fields like medical implants and diagnostic devices.
In addition to that, here are some other advantages:
- Biodegradable & Non Toxic
- Reusable & Recyclable
- Durable
- Rechargeable
- No Leakage & Overheating
- Very Light Weight & Flexible
7. Applications

Paper batteries have a broad spectrum of potential applications due to their unique properties. In the realm of consumer electronics, they can be used to power smartphones, laptops, and other handheld devices, paving the way for more lightweight and flexible designs. In the medical field, their biocompatibility and ability to operate using biological fluids can be harnessed for the operation of medical implants or biosensors. These batteries can be employed in smart packaging as well, where they can power sensors to monitor conditions like temperature or humidity. Furthermore, due to their low weight and high energy density, they are ideal for use in drones or other small, unmanned aerial vehicles. Lastly, given their environmental friendliness, they could also replace conventional batteries in various applications, contributing to the reduction of electronic waste and the promotion of green technology.
Here are some other applications for paper batteries:
- Low battery power devices like in calculators, wrist watch and other low drain devices
- Laptop batteries, mobile phones, handheld digital cameras
- Wireless communication devices like mouses, keyboard, bluetooth headphones,etc
Usage in Portable Electronic Devices
The advent of paper batteries could revolutionize the world of portable electronic devices. Their ultra-thin, lightweight, and flexible characteristics make them an excellent power source for devices like smartphones, tablets, and laptops. With these batteries, device designers won’t be restricted by the need to create space for a rigid, heavy battery component. Instead, the paper battery can be designed to fit seamlessly into the structure of the device, potentially leading to slimmer, more flexible gadgets. Moreover, their high power density means these devices could potentially operate for longer periods without needing to recharge, enhancing the user experience. Eco-conscious consumers would also appreciate their biodegradability, presenting a more sustainable alternative to traditional batteries.
Potential Roles in Renewable Energy Systems
Paper batteries could play a pivotal role in the renewable energy sector. One of the main challenges with renewable energy sources like solar and wind is their intermittent nature. For instance, solar panels do not generate electricity when there’s no sunlight and wind turbines stand still in calm weather. Paper batteries, with their high energy density and fast charging capabilities, could be used to store energy when production exceeds demand and release it when there’s a shortage. Their biodegradability also aligns well with the sustainability goals of renewable energy systems. Furthermore, the lightweight and flexible characteristics of paper batteries make them suitable for integration with portable or flexible solar panels, opening up new possibilities for the design and application of renewable energy systems.
Future Possibilities in Various Sectors
Looking ahead, the potential applications of paper batteries stretch far beyond renewable energy systems. In the medical field, these batteries could revolutionize the design and functionality of wearable devices and implants, allowing for a new generation of flexible, lightweight, and biodegradable medical gadgets. In the automotive industry, the high energy density and rapid charging capabilities of paper batteries could contribute to the development of more efficient electric vehicles. In the realm of consumer electronics, imagine smartphones, laptops, or wearables that are lighter, thinner, and more eco-friendly. Finally, considering their biodegradability and flexibility, paper batteries could also find applications in the development of sustainable smart cities, powering everything from smart sensors to public lighting. In conclusion, the possibilities are as vast and varied as the sectors that could benefit from this innovative technology.
Conclusion
In essence, paper batteries represent a significant stride forward in energy storage technology, offering a compelling blend of portability, efficiency, and ecological consciousness. Their high energy density, rapid charging, and biodegradable properties not only serve the needs of our technologically-driven society but also echo the global call for environmentally-friendly solutions. From renewable energy systems to consumer electronics and smart city applications, the utilization of paper batteries holds immense promise. However, as with any emerging technology, the road to widespread adoption will require further research, optimization, and rigorous testing. Nevertheless, the future of paper batteries looks bright and the world stands to gain immensely from its broad-ranging applications.










